From healthy and functional ingredient to health-giving product



Research journals, trade press and even newspaper headlines, regularly promote the health benefits of specific plants, compounds, etc. but converting a promising or functional ingredient, into a health-giving product that can be successfully marketed is not a straightforward process. Here, we explain the challenges – and possible solutions – involved.

Any successful food product or supplement has to meet customer expectations for looks, taste, texture etc. And, of course, it has to meet the highest food safety standards. But when you are working with a functional ingredient that has a known health benefit, you also have to make sure that the benefit is maintained throughout processing and shelf-life. That starts with investigating the physical and chemical properties of your ingredient – such as its solubility or response to heat, pH and other possible processing conditions.
There are many different possible application formats: powders, drinks, energy bars and so on. Each one has a different matrix and processing conditions that can impact the efficacy of the ingredient. It is vital to match the properties of your functional ingredient to an appropriate application. For example, a fat-soluble ingredient would work best in an emulsion (such as a dairy-like drink) or a fat-continuous system like chocolate. In contrast, water-soluble ingredients may be better in something like a sports drink.
Once you have matched the ingredient to an appropriate application, you then need to look at the dosing required to achieve a beneficial effect. Can you deliver enough of your ingredient via your chosen format in a way that fits into a typical diet? Consumers don’t want to have to drink 10 pots of yoghurt a day to see a benefit! And, of course, you need to validate the efficacy of your final product through lab studies, clinical trials or even through consumers tracking relevant biomarkers themselves using modern healthcare sensors.
Let’s take plant proteins as an example. For many people, sustainability is a key driver for reducing the amount of animal protein in their diet. At the same time, the health benefits of eating plant-based proteins are clearly supported in the literature. Replacing some animal protein with plant protein can potentially reduce blood pressure slightly, lower LDL (popularly called “bad”) cholesterol and help people with diabetes maintain glycaemic control. Plant-protein ingredients also typically include fibres and micronutrients that can promote gut health – and so overall health.
However, plant proteins come with many challenges. Not many plant sources provide all seven of the essential amino acids that people need. They also often come with anti-nutritional compounds which can, for example, inhibit the absorption of amino acids into the bloodstream.
Plant proteins are also typically less soluble than animal proteins, which affects both their digestibility and the potential sensory experience of eating products based on them. For example, trying to create high-protein, plant-based products in an aqueous environment – such as yoghurts, milks and other beverages – can lead to a gritty mouthfeel. Solubility is also key to the technical functionality of plant proteins and products based on them. Properties such as emulsification, foaming and gelling are often much lower in plant-based products than dairy-based equivalents.
Moreover, consumers in the Western world are not used to the flavours of plant-based proteins and often perceive them as “off-flavours”. Volatile compounds such as hexanal and methoxypyrazines can be quite dominant, leading to prominent beany- or pea-like flavours in proteins derived from legumes. Other sensory defects such as astringency and extreme dryness of the tongue are also often observed in plant proteins where a more-creamy mouthfeel would be wanted, such as in yoghurts.
All these issues can be addressed through smart extraction and processing choices, careful product design or a combination of both. For example, mild extraction and drying techniques have been shown to improve both the solubility and flavour profile of a protein ingredient. And you can combine different types of plant-based source such as legumes and cereals or rice into one product to give a complete amino acid profile.
Fermentation is a technology with a lot of potential to improve plant-based products. Through “bio-purification”, fermentation can remove unwanted molecules from ingredients – whether that is the volatile compounds that cause off flavours or the non-volatile compounds that have anti-nutritional effects. It can also be used to improve the texture and shelf life of products, or to add complex favour profiles through adjunct cultures of final products. Of course, even a technique as versatile as fermentation has to be carefully matched to the target application. Using fermentation to prevent spoilage via acidification would be a good option for, say, a yoghurt-based drink but not for something like a milk-type beverage.
There are many tools for validating health benefits of foods and supplements such as in vitro digestion models and cell lines that mimic the body parts or systems such as the gut wall, muscle fibres or the immune system. At NIZO, we have also developed a miniature fermentation system that mimics the gut microbiome. Used together, these tools enable lab-based studies that give you early indication of whether the compounds responsible for your desired health benefits will survive the gastrointestinal environment – for example can your novel plant protein or combination of proteins b digested. Clinical studies with healthy volunteers can then provide evidence of the compound passing into the bloodstream and whether it does actually have an effect on the human body.
Combining different in vitro models can give you the deeper insight to understand what will really happen when your product is ingested. For example, in dairy, it is well known that levels of essential amino acids in the blood rise quicker after ingesting whey protein than casein even though the amino acid composition of the two is quite similar. Gastric model studies reveal aggregate formation for casein. Because the larger particles formed by this aggregation delay gastric emptying, the amino acids from casein appear later in the bloodstream.
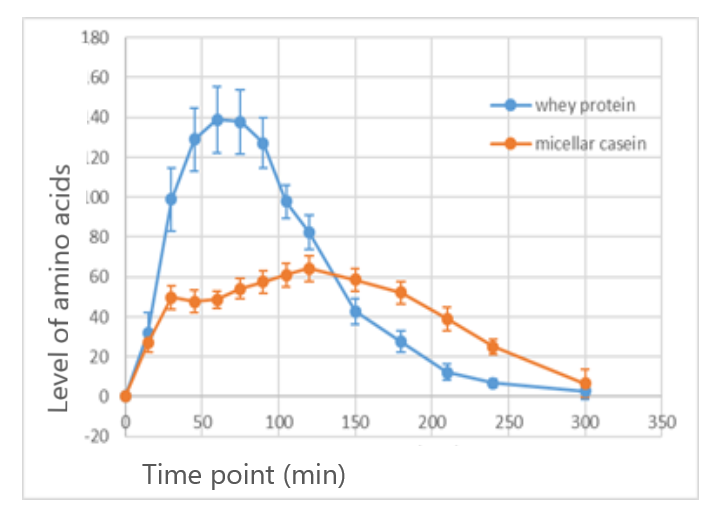
The same tools can be used to investigate amino acid absorption from plant-based products, but here the picture is more variable. Plant-based products haven’t yet reached nutritional parity with animal-based protein – which is the goal for upcoming product generations. But, as mentioned above, we do know that the source, processing, and product matrix play a big role. So, it is important to carry out health benefit studies alongside product development, and feed the insight from those studies back to optimize the product as you go.